Long-term protection against SARS-CoV-2
Edgar Meinl and his team show that SARS-CoV-2-recognizing memory B cells circulate in the blood even after loss of specific antibodies.
As we enter the third year of the COVID-19 pandemic, one of the central questions continues to be whether and how a long-term immune protection can be achieved after infection or vaccination.
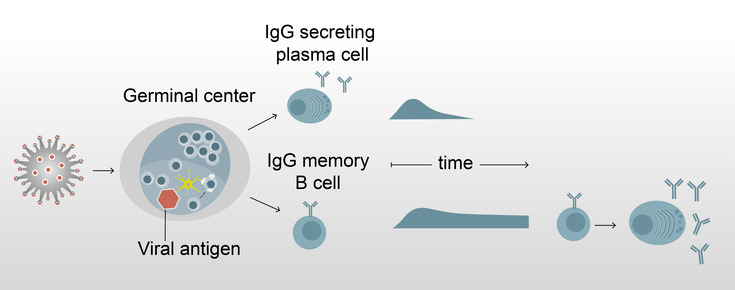
In principle, the adaptive immune system can protect against re-infection or disease caused by the same pathogen. The effect of vaccinations is in fact based on this ability. The underlying defense reaction requires the interaction of T cells, B cells and antibodies produced by B cells. For the development of "high-affinity" (highly effective) antibodies, the maturation of B cells in so-called germinal centers in lymphoid organs such as the lymph nodes and the spleen is crucial. Two types of B cells leave the germinal centers, antibody-producing plasma cells and memory B cells.
While some COVID-19 patients still have antibodies in their blood 6-9 months after infection, others lose their specific antibodies. Edgar Meinl's research group at the Biomedical Center has now shown that memory B cells remain in the blood after infection with SARS-CoV-2, even when antibodies are no longer detectable. To study these memory B cells, the group differentiated B cells derived from blood into antibody-producing cells in cell culture and further analyzed the produced antibodies. Blood cells from a total of 17 already recovered COVID-19 patients were examined. In five of them, specific antibodies were no longer detectable in the blood as early as 5-8 months after infection.
In all patients, Edgar Meinl and his team found SARS-CoV-2-specific memory B cells that produced antibodies of the type immunoglobulin-G (IgG), and in 11 of the 17 patients also those that produced antibodies of the type immunoglobulin-A (IgA). These antibodies blocked the binding of the viral receptor binding domain to the cellular receptor ACE-2 and had neutralizing effects against infectious viruses in cell culture, as shown in cooperation with Prof. Oliver Keppler of the Max von Pettenkofer Institute, Virology, LMU.
The work thus shows that functional SARS-CoV-2-specific memory B cells are detectable in the blood for a long time after COVID-19 infection. This finding is of critical importance to the question of long-term immunity, as memory B cells can rapidly differentiate into antibody-producing cells upon re-infection (or infection after vaccination) and also evolve to better bind viral variants. The results of Edgar Meinl's team also show that for the detection of a passed infection, the examination of memory B cells in cell culture may be more suitable than the analysis of antibodies in the blood, since these cells, in contrast to the antibodies, are preserved over a longer period of time.
Publication: Winklmeier et al.: Persistence of functional memory B cells recognizing SARS-CoV-2 variants despite loss of specific IgG, iScience 2022

